 Pheromonal communication often involves providing information about the reproductive status of a potential mate. Many species respond to chemical messages, known as pheromones, sent by another individual (Wysocki & Preti, 2004). Dogs’ extraordinary olfactory abilities may be due to the increased number of functional genes for olfactory receptors (between 8), compared to the fewer than 400 observed in humans and other primates (Niimura & Nei, 2007). There is some evidence to suggest that dogs can “smell” dangerous drops in blood glucose levels as well as cancerous tumors (Wells, 2010). We often think of dogs as having far superior olfactory systems than our own, and indeed, dogs can do some remarkable things with their noses. There is tremendous variation in the sensitivity of the olfactory systems of different species. Olfactory receptors are the hair-like parts that extend from the olfactory bulb into the mucous membrane of the nasal cavity. From the olfactory bulb, information is sent to regions of the limbic system and to the primary olfactory cortex, which is located very near the gustatory cortex (Lodovichi & Belluscio, 2012 Spors et al., 2013). Once an odor molecule has bound a given receptor, chemical changes within the cell result in signals being sent to the olfactory bulb: a bulb-like structure at the tip of the frontal lobe where the olfactory nerves begin. Small hair-like extensions from these receptors serve as the sites for odor molecules dissolved in the mucus to interact with chemical receptors located on these extensions ( Figure). Olfactory receptor cells are located in a mucous membrane at the top of the nose. (credit a: modification of work by Jonas Töle credit b: scale-bar data from Matt Russell) Smell (Olfaction) 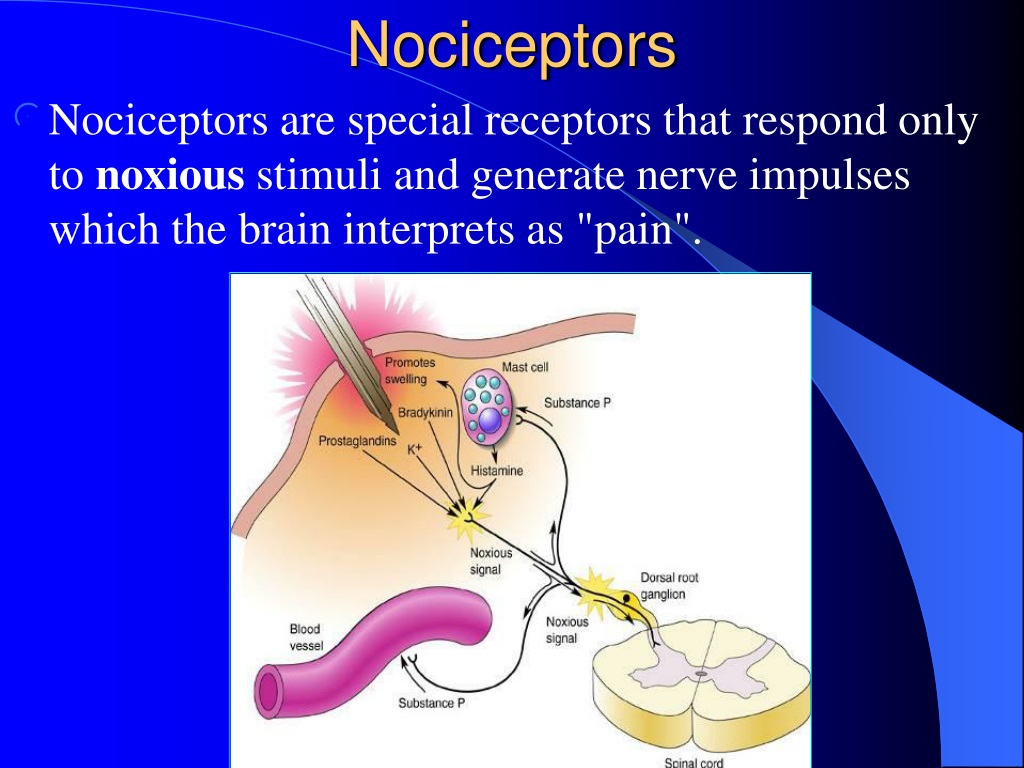
(b) This micrograph shows a close-up view of the tongue’s surface. (a) Taste buds are composed of a number of individual taste receptors cells that transmit information to nerves. Taste information is transmitted to the medulla, thalamus, and limbic system, and to the gustatory cortex, which is tucked underneath the overlap between the frontal and temporal lobes (Maffei, Haley, & Fontanini, 2012 Roper, 2013). Taste molecules bind to receptors on this extension and cause chemical changes within the sensory cell that result in neural impulses being transmitted to the brain via different nerves, depending on where the receptor is located. Taste buds have a life cycle of ten days to two weeks, so even destroying some by burning your tongue won’t have any long-term effect they just grow right back. Taste buds are formed by groupings of taste receptor cells with hair-like extensions that protrude into the central pore of the taste bud ( Figure). Molecules from the food and beverages we consume dissolve in our saliva and interact with taste receptors on our tongue and in our mouth and throat. There is also a growing body of experimental evidence suggesting that we possess a taste for the fatty content of a given food (Mizushige, Inoue, & Fushiki, 2007). Umami is actually a Japanese word that roughly translates to yummy, and it is associated with a taste for monosodium glutamate (Kinnamon & Vandenbeuch, 2009). Research demonstrates, however, that we have at least six taste groupings. You have learned since elementary school that there are four basic groupings of taste: sweet, salty, sour, and bitter. 
For example, when we describe the flavor of a given food, we are really referring to both gustatory and olfactory properties of the food working in combination.  
There is a pronounced interaction between our chemical senses. Our study greatly expands the molecular characterization of C-LTMRs and suggests that this particular population of neurons shares some molecular features with A beta and A delta low-threshold mechanoreceptors.Taste (gustation) and smell (olfaction) are called chemical senses because both have sensory receptors that respond to molecules in the food we eat or in the air we breathe. We validated 48 MRGPD- and 68 C-LTMRs-enriched genes using a triple-staining approach, and the Ca(v)3.3 channel, found to be exclusively expressed in C-LTMRs, was validated using electrophysiology. Cluster analysis of RNA-seq profiles of the purified neuronal subsets revealed 486 and 549 genes differentially expressed in MRGPRD-expressing neurons and C-LTMRs, respectively. Here, we used a mouse model that genetically marks C-LTMRs and MRGPRD(+) neurons in combination with fluorescent cell surface labeling, flow cytometry, and RNA deep-sequencing technology (RNA-seq). The molecular mechanisms underlying these diametrically opposite functions are unknown. Cutaneous C-unmyelinated MRGPRD(+) free nerve endings and C-LTMRs innervating hair follicles convey two opposite aspects of touch sensation: a sensation of pain and a sensation of pleasant touch. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
